Loss of Cortical SSEP Due To Loss of Receptor Activation
Earlier in this 30 Days of Neuromonitoring series, I wrote about cortical and subcortical reorganization after a loss of afferent feedback from carpal tunnel syndrome and how that affects intraoperative somatosensory evoked potentials. To follow along those same lines, I’d like to reference another study by (Valeriani, 1996) titled Central nervous system modifications in patients with lesion of the anterior cruciate ligament of the knee (similar topic, but this one deals with the receptor).
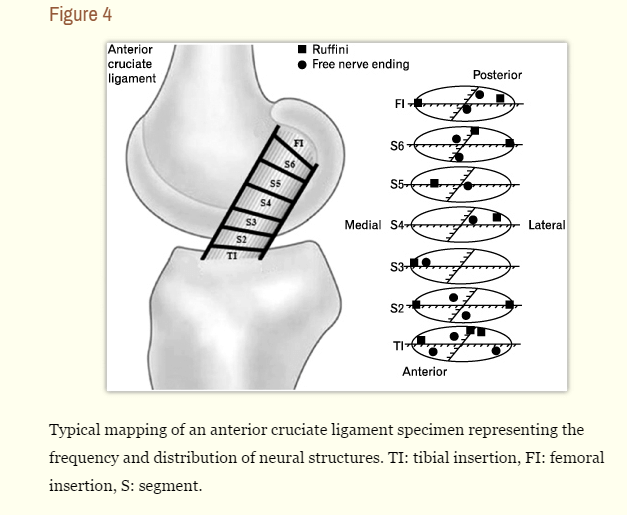
In this study, they again looked at the effects that loss of afferent information has on our central nervous system and our ability to record physiologic responses. In this case, they studied patients with loss of proprioception in the knee after injury to the anterior cruciate ligament. Their 2 major finding were the following:
- The common peroneal nerve showed loss of cortical SSEPs, but no real loss in subcortical and spinal amplitudes. In the diagnostic world, that usually means something causing a conduction block along the medial lemniscus. In the intraoperative neuromonitoring world, we tend to think conduction block in medial lemniscus due to preexisting lesion, pre-existing ischemic changes (which we can sometimes jump the gun a little bit on) or anesthetic effects. But their second finding eliminated those options.
- These same findings were not seen with stimulation to the posterior tibial nerve at the knee or ankle, nor the peroneal nerve in the lower leg. They were able to record cortical, subcortical and spinal SSEP from these more distal stimulating sites. This now makes a medial lemniscus conduction block or lesion very low on the probability scale and points to something cortical in nature.
ACL Injury Vs. Carpal Tunnel Syndrome As It Relates To SEP
What we are seeing here is not a loss of conduction of the afferent information, as was discovered with the carpal tunnel group, but a loss of the summation from the receptors themselves. The injury caused damage to the receptor and decreased the day-to-day barrage of proprioceptive input that drives our central nervous system.
Since a small but significant portion of the IONM community is made up of chiropractors, physical and occupational therapist, exercise physiologist and other healthcare providers who come from a receptor-based therapy background, this shouldn’t be too shocking. In the application and recording of physical medicine, we look to ramp up areas of deficient afferent input through direct or indirect means.
It’s a leading explanation as to why exercise has one of the best track records for curing depression, or how mirror therapy works as a visual feedback therapy for stroke patients, etc.
This is just the measurement of the reverse happening. Loss of receptor activation – leads to loss of central neuronal activation – leads to reorganization of surrounding structures as a means to cope.
Enough Chiropractic – How Might This Be Useful In Neuromonitoring?
I had four major thoughts from this paper:
- The ability to feel a cotton swab or pin prick is not the only way to assess sensory afferents. Proprioception and 2-point discrimination are also of value when recording somatosensory evoked potentials. This could be an additional reason as to why some patients have poor SSEP even though they can “feel” everything.
- In any patient with a history of loss of afferent input, alternative stimulating locations need to be planned for ahead of time. Always be planning for optimization.
- This paper only showed cortical changes, while the carpal tunnel had both cortical and subcortical changes. Is the degree of loss of afferent firing the cause?
- This paper and the carpal tunnel paper showed 2 different ways SSEP can be affected by a loss of afferent input. They both showed to have reorganization over the areas specific to the somatotopic representation of that anatomical area with an afferent deficit. Might we carry this over to other common scenarios that we encounter as surgical neurophysiologist (you should be thinking spinal injury here)? While it is well accepted that the muscles that control fine movement have a higher density of muscle spindles, it might not be widely appreciated as to the amount in the cervical spine versus other areas (see numbers below). What about a loss of afferent input from the cervical spine for our MVA patients? While we aren’t running an SSEP off of a cervical nerve root or anything, the sheer amount of afferent input might affect the overall cortical drive, and in return smaller cortical SSEP overall. (Sorry, some more chiropractic thoughts: this last one is a theory, still looking for a paper for some validity. But I know that chiropractors would love to see something like this. Lowered cortical output due to spinal injury >>> manipulation of the spine causing a fast stretch to muscles, possibly resetting the gain of the muscle spindle cells >>> improved afferent feedback >>> improved SSEP).
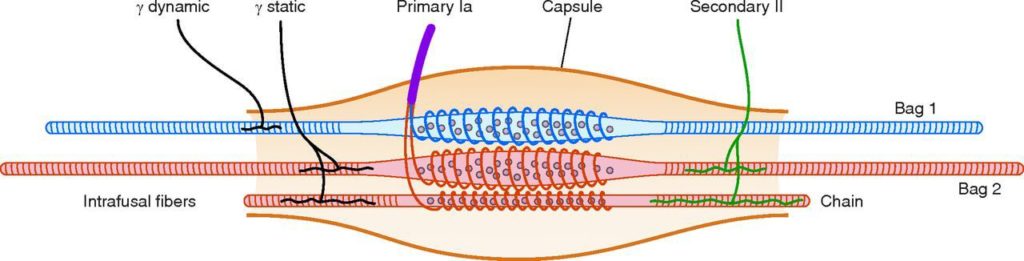
Density of Muscle Spindles Per Gram Of Muscle Tissue
Inferior Oblique 242
Superior Oblique 190
Rectus Capitis Posterior Major 98
Rectus Capitis Posterior Minor 98
Lateral Pterygoid 20.3
Opponens Pollicis 17.3
First Lumbrical 16.5
Trapezius 2.2
Latissimus Dorsi 1.4
Medial Pterygoid 0
Does anyone else have another take on this article as it relates to intraoperative neuromonitoring that I didn’t consider? I always like to hear other people’s perspective on things, especially when it isn’t exactly a straight line to come to some sort of conclusion. There isn’t much talk about the effect of the receptor on our SSEP, especially considering the fact that we bypass the receptors with electric current directly stimulating the nerve.
Just place your comment in the section below.

Joe Hartman
Subscribe to our Awesome Newsletter.
Keep Learning
Here are some related guides and posts that you might enjoy next.
How To Have Deep Dive Neuromonitoring Conversations That Pays Off…
How To Have A Neuromonitoring Discussion One of the reasons for starting this website was to make sure I was part of the neuromonitoring conversation. It was a decision I made early in my career... and I'm glad I did. Hearing the different perspectives and experiences...
Intraoperative EMG: Referential or Bipolar?
Recording Electrodes For EMG in the Operating Room: Referential or Bipolar? If your IONM manager walked into the OR in the middle of your case, took a look at your intraoperative EMG traces and started questioning your setup, could you defend yourself? I try to do...
BAER During MVD Surgery: A New Protocol?
BAER (Brainstem Auditory Evoked Potentials) During Microvascular Decompression Surgery You might remember when I was complaining about using ABR in the operating room and how to adjust the click polarity to help obtain a more reliable BAER. But my first gripe, having...
Bye-Bye Neuromonitoring Forum
Goodbye To The Neuromonitoring Forum One area of the website that I thought had the most potential to be an asset for the IONM community was the neuromonitoring forum. But it has been several months now and it is still a complete ghost town. I'm honestly not too...
EMG Nerve Monitoring During Minimally Invasive Fusion of the Sacroiliac Joint
Minimally Invasive Fusion of the Sacroiliac Joint Using EMG Nerve Monitoring EMG nerve monitoring in lumbar surgery makes up a large percentage of cases monitored every year. Using EMG nerve monitoring during SI joint fusions seems to be less utilized, even though the...
Physical Exam Scope Of Practice For The Surgical Neurophysiologist
SNP's Performing A Physical Exam: Who Should Do It And Who Shouldn't... Before any case is monitored, all pertinent patient history, signs, symptoms, physical exam findings and diagnostics should be gathered, documented and relayed to any oversight physician that may...





0 Comments