Is SSEP Improvement Sensitive For Detecting Complete Decompression Of The Spinal Cord Or Nerve Root?
In an article that I wrote years ago, titled Brainstem Auditory Evoked Potential Used To Predict The Need For Duraplasty In Chiari Malformation Type I Decompression, I described some of my skepticism of the technique (since that time, I have done more of those cases and have written an update to that post). A lot of that skepticism comes from what we know about how the spinal cord and peripheral nerves act during times of subacute or chronic compression and trying to apply what’s known about SSEP improvement to ABR improvement.
Stories have surfaced about people in the field telling surgeons that they have seen improvements in their SSEP signals from baseline data after decompression of the spinal cord or nerve roots in order to assist in determining if decompression was adequate. And it’s happening on a routine basis. So I wanted to address the topic specifically.
But before I do, and before you go on to read the rest of this post, answer this question…
SSEP Improvement In The Literature
While some of the SSEP improvements might be subtle and easily missed, misunderstood or misinterpreted, this should be considered irregular findings. But there are those cases where a sudden change happens after decompression. If it is a gradual increase, you really need to consider some other cause.
In fact you have to really dig to find examples that might show claims of improved SSEP signals after decompression of the spine, improved SSEP in patients with a syrinx , increase in SSEP amplitude after draining a spinal cyst, SSEP improvement seen more with when decompression of the lateral recess was performed along with a discectomy over discectomy alone (although it had no clinical significance postoperatively), SSEP latency and amplitude improvements seen intraoperative, 3 weeks and 3 months have been associated with improved postoperative results or even SSEP improvement after removal of a parietal tumor.
But after reading those articles, you’re left having this conversation in your head…

“I see that it is possible and it is happening, but what significance does it have?”
If an SSEP loses 50% of its amplitude, or a tcMEP loses 100% of its amplitude, I have a pretty good understanding of what that means for the patient. If I see an SSEP amplitude improvement of 5% or 50% after decompression of a lumbar nerve root or removal of a spinal tumor, I have no idea what that means for the patient. At this point in time, all I can do is make assumptions (again, there seems to be a little more progress with ABR and SSEP with Arnold Chiari malformation bony decompression, but not there yet).
What do we need to consider when we do see improvement in SSEP?
So all you are left with are assumptions. But those still might play some role in a medical decision being made when we consider what we feel it “should” mean.
If you have an acute injury in the spinal cord or nerve root and see no improvement, it is probably best not use that information as diagnostic criteria to determine if further decompression is warranted.
Also, should there be a sudden improvement in SSEP after decompression of the spinal cord or nerve root, this does not signify that any further decompression is needed. Other criteria used by the surgeon should be used to determine the level of decompression.
If the surgeon is at a point in the procedure where they need to weight the risk of surgical options going forward, knowing information about SSEP improvement might help them in their medical decision. We see this when deciding to perform duraplasty after bony decompression during surgery for a Chiari malformation (which has its own risk and no proof in additional benefit), and similar reasoning may be adopted to other surgical scenarios.
Why might we be hesitant to use SSEP improvement for decompression of the spinal cord or nerve root for diagnostic purposes?
Reason #1 – The Peripheral Nerve Response To Chronic Compression And/Or Tension
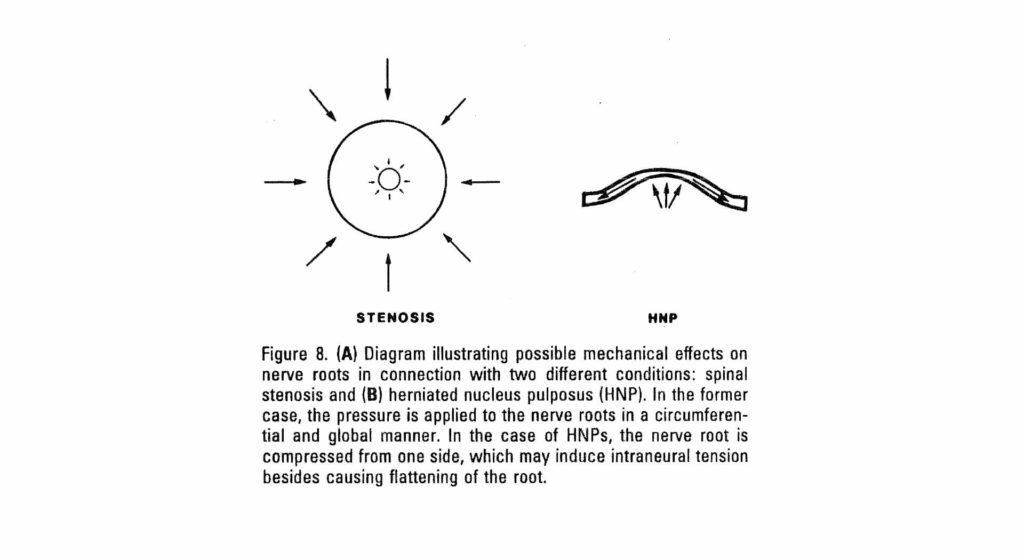
The patient population that we see in spine surgery largely have the diagnosis of stenosis, radiculopathy, and myelopathy. In a high percentage of those patients, this has developed over a long period of time and affects only the large diameter fibers.
In the peripheral nerves, we know that it is the largest diameter nerves that are affected first by compressive forces. These are the very same axons that are utilized in SSEPs. So it is safe to say that these are the fibers that should show changes in conduction. And If this happens for too long, then the axons being crushed or stretched will degenerate.
Smaller fibers that carry pain (C fibers) are usually spared in compressive injuries up to the point where the force applied obliterates the large diameter fibers. This is because the amount of force needed to displace the viscous contents of the narrower diameter tubules is greater than the amount of force needed to crush the larger diameter nerve fibers.
Should surgical intervention take place before Wallerian degeneration happens, the permanent damage done to these fibers can affect the transmission of electrical activity. The myelin sheath will react to the combination of the compressive forces applied and restricted movement of the fibers under the area of compression causing tension or tethering (normal range of 2-5 mm of movement in nerve roots within the neuroforamen with straight leg raises). What you might end up getting is a deformation of the myelin sheath over the axon. Though this has been studied in median nerve entrapment syndromes, it has been theorized to take place in other areas of compression in the peripheral nervous system.
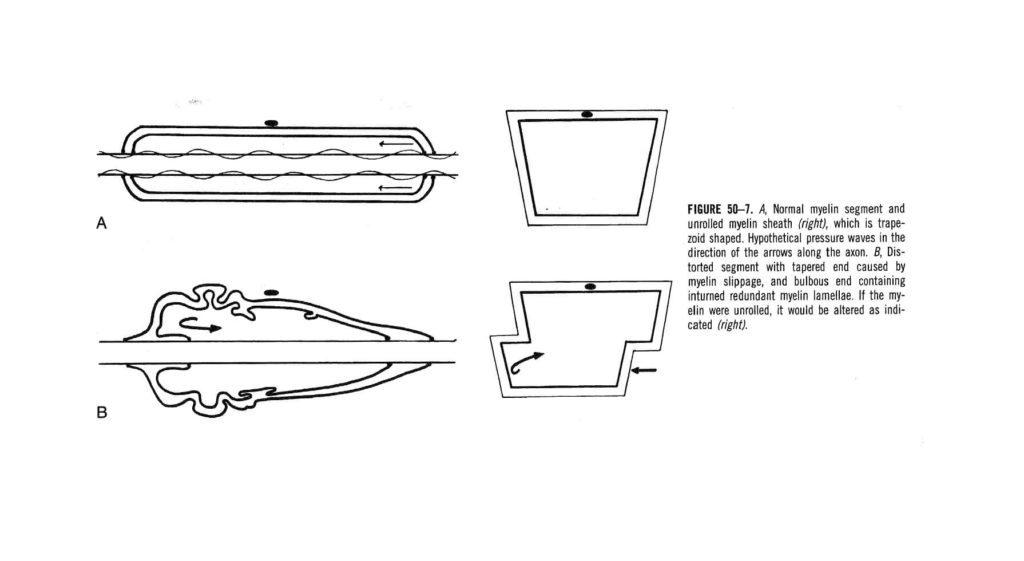
This disruption the nerve’s architecture can result in changes in transmission of electrical pules that will not change with a sudden release of compression or tension after surgical intervention.
Reason #2 – The Ability Of SSEP To Detect Early Structural Loss, But Inability To Differentiate It From Functional Loss. And The Opposite During Recovery.
Acute loss of somatosensory evoked potentials is a sensitive test to acute tension or compression placed on the conducting pathways and can lead to permanent damage. In the surgical setting, reversal of maneuvers, if possible, is suggested in order to prevent injury. The rate of return of the SSEP potentials can be of some value in predicting poor neurological outcomes if the potentials do not return within 30 minutes to a couple hours. On the other hand, rapid recovery does not guarantee that there has not been some significant axonal damage even though functional outcomes are usually positive. The nervous system is able to adapt and provide little to no apparent changes to function even under extreme structural damage.
PRO TIP

“Read the last sentence again. Significant structural damage can happen in the absence of functional reduction. We need to really question some of these papers and the rates of “false positives” being claimed.”
- Because the patient fully recovers does not mean that the change in SSEP was meaningless. It also does not mean that the nervous tissue has been destroyed.
- Acute loss of SSEP is a much better indicator of acute axonal distress than it is for chronic axonal destruction.
Understanding this concept is important in realizing that SSEP is better from a preventative standpoint that predictive.
In chronic myelopathy, even in cases where the exact timing of the initial injury is known (for example, car accident), a number of variables to the extent of cord injury can make your head explode.
- Did this patient sneeze too hard and cause additional trauma to the compressed cord?
- Did this patient have times of low blood flow causing transient ischemia?
- Did this patient have a pre-existing injury to the cord?
We don’t know where they are on the timeline of ongoing injury and recovery to conducting fibers, nor the capability of the other conducting fibers to preserve function in light of a severe injury.
In the (Blight, 1990) paper, they showed that significant acute damage to conducting fibers in the spinal cord (87-99% loss of myelinated axons in white matter) demonstrated variable responses in SSEP recordings in the short, intermediate and long-term.
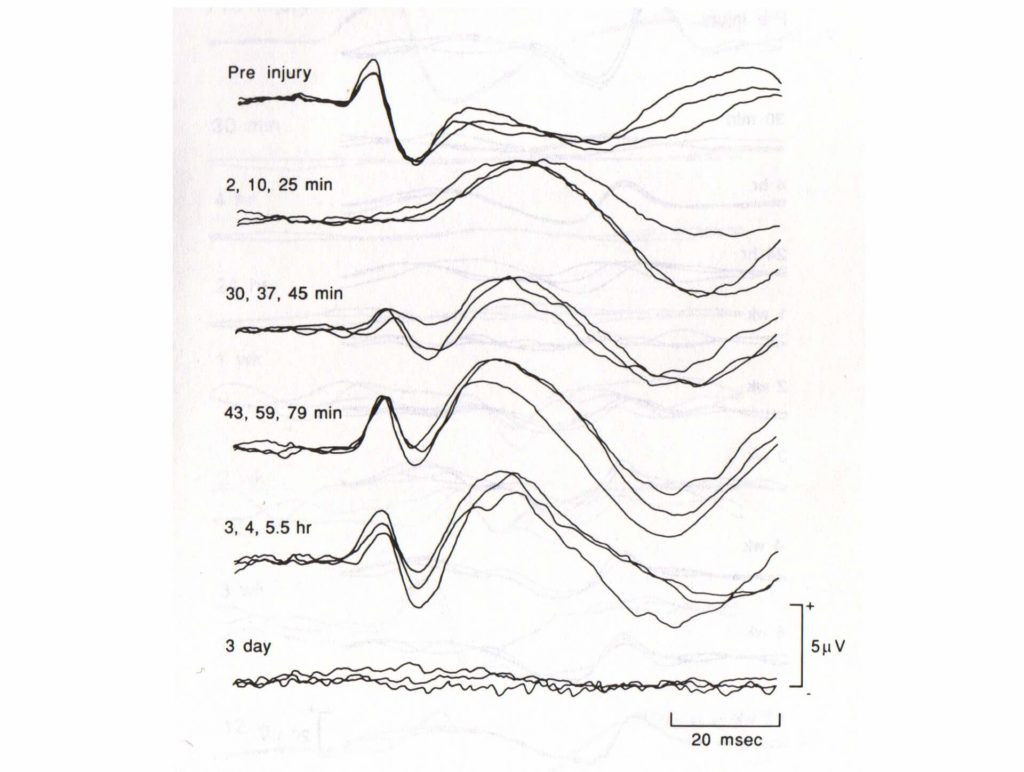
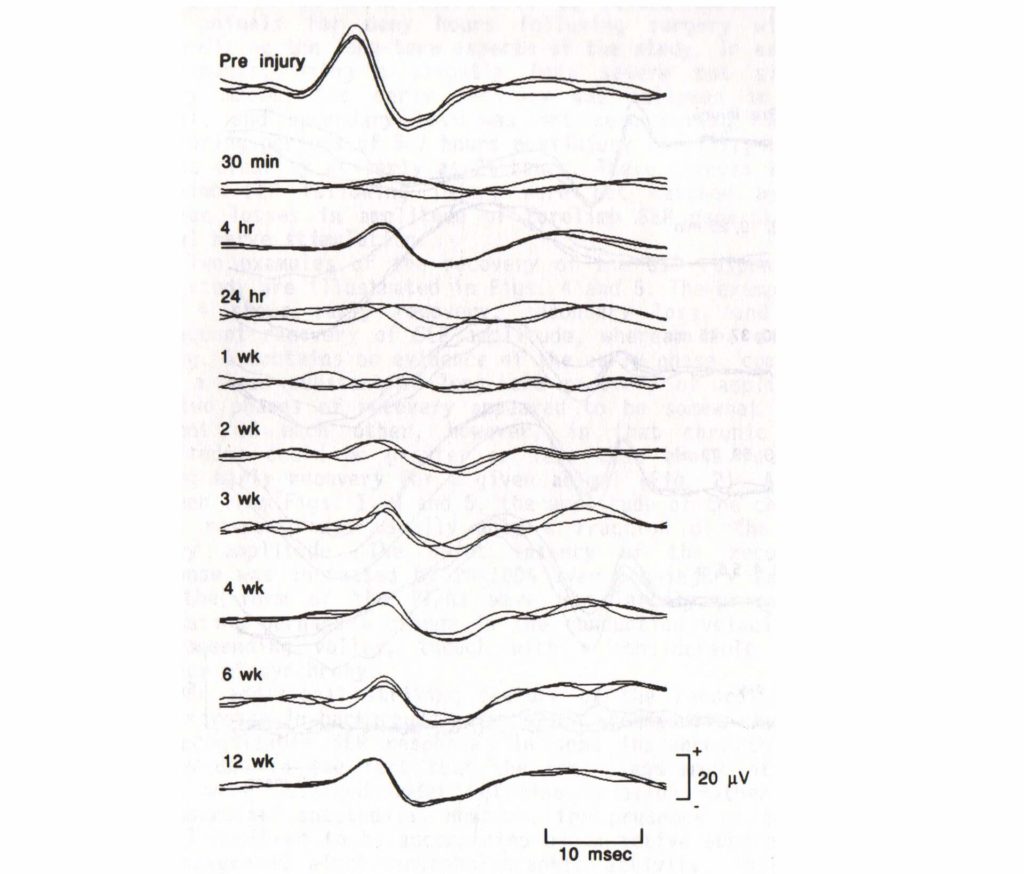
Here are 3 examples of SSEP recordings after a controlled traumatic event was performed on the spinal cord in anesthetized cats. The potentials demonstrated 3 very different outcomes.
#1 – Immediate SSEP loss – SSEP improvement – secondary loss in the long-term
#2 – Immediate loss of signal – partial recovery hours later – secondary loss – partial recovery in the long-term
#3 – Immediate loss – Delayed recovery over long-term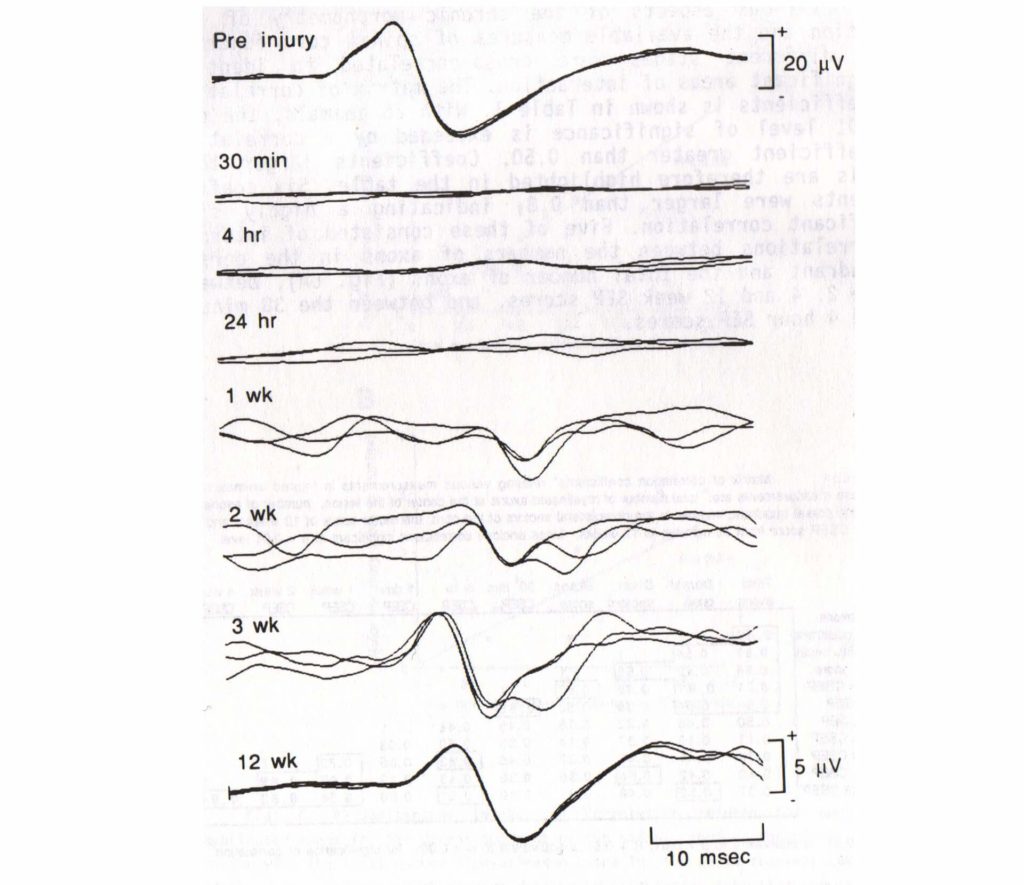
“So it looks like even though it can happen, SSEP improvement rarely does, and we don’t really have a good understanding of what it means. Ok, I’ll make you a deal. I’ll stop asking for SSEP improvement if you stop calling every single intraoperative EMG Deal?.”

Want new articles before they get published?
Subscribe to our Awesome Newsletter.
1 Comment
Keep Learning
Here are some related guides and posts that you might enjoy next.
How To Have Deep Dive Neuromonitoring Conversations That Pays Off…
How To Have A Neuromonitoring Discussion One of the reasons for starting this website was to make sure I was part of the neuromonitoring conversation. It was a decision I made early in my career... and I'm glad I did. Hearing the different perspectives and experiences...
Intraoperative EMG: Referential or Bipolar?
Recording Electrodes For EMG in the Operating Room: Referential or Bipolar? If your IONM manager walked into the OR in the middle of your case, took a look at your intraoperative EMG traces and started questioning your setup, could you defend yourself? I try to do...
BAER During MVD Surgery: A New Protocol?
BAER (Brainstem Auditory Evoked Potentials) During Microvascular Decompression Surgery You might remember when I was complaining about using ABR in the operating room and how to adjust the click polarity to help obtain a more reliable BAER. But my first gripe, having...
Bye-Bye Neuromonitoring Forum
Goodbye To The Neuromonitoring Forum One area of the website that I thought had the most potential to be an asset for the IONM community was the neuromonitoring forum. But it has been several months now and it is still a complete ghost town. I'm honestly not too...
EMG Nerve Monitoring During Minimally Invasive Fusion of the Sacroiliac Joint
Minimally Invasive Fusion of the Sacroiliac Joint Using EMG Nerve Monitoring EMG nerve monitoring in lumbar surgery makes up a large percentage of cases monitored every year. Using EMG nerve monitoring during SI joint fusions seems to be less utilized, even though the...
Physical Exam Scope Of Practice For The Surgical Neurophysiologist
SNP's Performing A Physical Exam: Who Should Do It And Who Shouldn't... Before any case is monitored, all pertinent patient history, signs, symptoms, physical exam findings and diagnostics should be gathered, documented and relayed to any oversight physician that may...
Help Spread The Word. Social Media Works!





Thank you. I’m sure we’ve all had this question